






|
Leeper Group | ||||
| | |||||

|

|
 |
 |
 |
|
  | ||||||
|
Abstract 62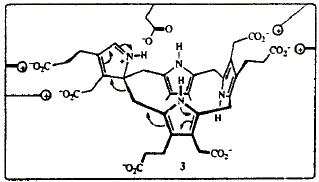 F. J. Leeper
F. J. Leeper "Evidence for a Spirocyclic Intermediate in the Formation of Urogen III by Cosynthetase" in The Biosynthesis of the Tetrapyrrole Pigments, 1994, J. Wiley & Sons, Chichester, p. 111-123. Full Text In the course of the cyclization of the linear tetrapyrrole hydroxymethylbilane to uroporphyrinogen III, catalysed by uroporphyrinogen III synthase (cosynthase), ring D of the bilane becomes inverted. Many different mechanisms have been proposed for this transformation but the most economical is one involving a spirocyclic pyrrolenine. Synthesis of a spirolactam, and other compounds closely related to the spirocyclic pyrrolenine, has shown that such compounds are not impossibly strained. The spirolactam is a powerful inhibitor of the enzyme, which suggests it does resemble an intermediate in the enzymic process. In the synthetic procedure to make an ester of the spirolactam the two products obtained were initially thought to be conformational isomers. However, molecular mechanics calculations on a model of the spirolactam predicted that several low energy conformations should exist and that the energy barriers for their interconversion are all lower than 32 kJ/mol. Reinvestigation revealed that one of the two products is in fact a macrocyclic dimer with a 28-membered ring. On the basis of the predicted preferred conformations of the spirolactam and of uroporphyrinogen III, a detailed three-dimensional mechanism is proposed, along with a rationalization of how the rearrangement of ring D may be directed by the enzyme.
|
||||||
  | ||||||