






|
Leeper Group | ||||
| | |||||

|

|
 |
 |
 |
|
  | ||||||
|
Abstract 38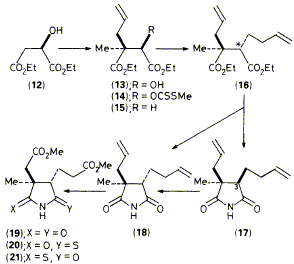
W. G. Whittingham, M. K. Ellis, P. Guerry, G. B. Henderson, B. Muller, D. A. Taylor, F. J. Leeper and A. R. Battersby "Syntheses Relevant to Vitamin B12 Biosynthesis: The Malate Route to (-)-Ring-B Imide and Synthesis of the 2,7,20-Trimethylisobacteriochlorin" J. Chem. Soc., Chem. Commun.,1989, 1116-1119. Full Text An efficient enantioselective synthesis of (-)-ring-B imide (19) from R-malic acid is outlined and this product is used for synthesis, by a photochemical route, of the 2,7,20-trimethylisobacteriochlorin (3), of importance for biosynthetic research on vitamin B12.
|
||||||
  | ||||||