






|
Leeper Group | ||||
| | |||||

|

|
 |
 |
 |
|
  | ||||||
|
Abstract 37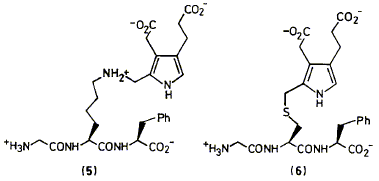
A. D. Miller, F. J. Leeper and A. R. Battersby "Biosynthesis of Porphyrins and Related Macrocycles, Part 34. Synthesis and Properties of S-Pyrrolylmethylcysteinyl and e-N-Pyrrolylmethyl-lysyl Peptides" J. Chem. Soc., Perkin Trans. 1,1989, 1943-1956. Full Text Syntheses are described of two compounds having a pyrrolylmethyl group attached respectively to the sulphur of cysteine (6) and the e-nitrogen of lysine (5). These compounds were built to act as model systems for two possible ways in which pyrrolylmethyl groups could be bound to the protein of the enzyme hydroxymethylbilane synthase (HMBS), also known as porphobilinogen deaminase. We show how results from these studies were important in the discovery and characterisation of the novel pyrromethane cofactor (4) of HMBS.
|
||||||
  | ||||||