






|
Leeper Group | ||||
| | |||||

|

|
 |
 |
 |
|
  | ||||||
|
Abstract 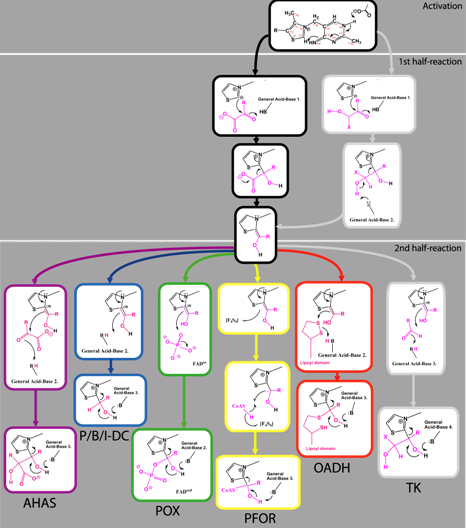 112 112René Frank, Finian Leeper and Ben F. Luisi, "Structure, mechanism and catalytic duality of thiamine-dependent enzymes." Cell. Molec. Life Sci., 2006, 64, 892-905. Full Text Thiamine is an essential cofactor that is required for processes of general metabolism amongst all organisms, and it is likely to have played a role in the earliest stages of the evolution of life. Here, we review from a structural aspect the enzymatic mechanisms that involve the cofactor and the origins or their specificity. We explore asymmetry within homo-dimeric thiamine diphosphate (ThDP)-dependent enzyme structures and discuss how this may be correlated with the kinetic properties of half-of-the-sites-reactivity, and negative cooperativity. It is likely these structural and kinetic hallmarks may arise through reciprocal coupling of active sites. This mode of communication between distant active sites is not unique to ThDP-dependent enzymes, but is widespread in other classes of oligomeric enzyme. Thus it appears likely that to be a general phenomenon reflecting a powerful mechanism of accelerating the rate of a chemical pathway. Finally, we speculate on the early evolutionary history of the cofactor and its ancient association with protein and RNA.
|
||||||
  | ||||||