






|
Leeper Group | ||||
| | |||||

|

|
 |
 |
 |
|
  | ||||||
|
Abstract 67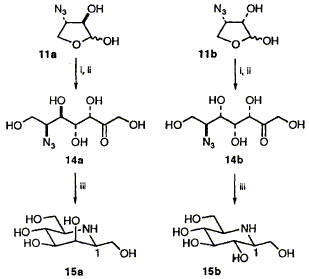 K. E. Holt, F. J. Leeper and S. Handa
K. E. Holt, F. J. Leeper and S. Handa "Synthesis of beta-1-Homonojirimycin and beta-1-Homomannojirimycin using the Enzyme Aldolase" J. Chem. Soc., Perkin Trans.1,1994, 231-234. Full Text The four stereoisomers of the four-carbon azido sugar 11 have been stereoselectively synthesised by a route involving Sharpless epoxidation and all are found to be substrates for rabbit muscle fructose 1,6-bisphosphate aldolase, giving (after treatment with phosphatase) 6-azido-6-deoxyheptuloses 14; hydrogenation of 14a and 14b gave beta-1-homomannojirimycin 15a and beta-1-homonojirimycin 15b with high selectivity.
|
||||||
  | ||||||